Bard PowerPort Catheter
Bard PowerPort Catheter Failure
Lawsuits Are Now Being Filed Against Becton, Dickinson, and Company; C.R. Bard, Inc.; Bard Access Systems, Inc. (“Bard), the Manufacturers of the Bard PowerPort Catheter. These Lawsuits Allege That Bard Failed to Warn Patients of the Dangers of the PowerPort Catheter, Failed to Modify the Product to Make it Safer, and Failed to Update Any Warning Labels. the Food and Drug Administration Has Not Issued an Active Recall.
Bard Has Even Attempted to Pass the Blame Onto Doctors, Saying That They Messed Up During Implantation, Clearly Attempting to Conceal the Dangers of Their Own Products. Bard Has Received Numerous Reports of Adverse Effects; However, Continues to Market and Sell the PowerPort Catheter as Safe.
What is a Bard PowerPort Catheter?
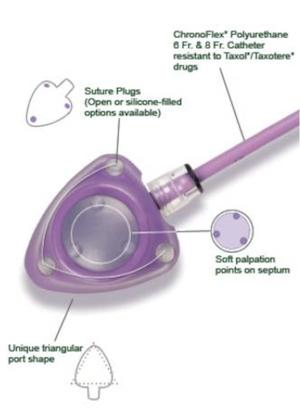
People Who Require Repeated Access to Their Vascular System—be it for Medication Infusions or Diagnostic Studies—will Sometimes Require a Permanent Catheter to Facilitate Fast Access When Necessary. They Are Frequently Used for Cancer Patients to Receive Chemotherapy. the Bard PowerPort Catheters Consist of Two Parts: an Injection Port Where the Medications or Fluids Are Administered and the Actual Bard PowerPort Catheter Implant That Stays Inside the Patient. the Injection Port is Not at Issue in the Bard PowerPort Lawsuits.
The Implanted Catheter is Designed From a Material Called “Chronoflex”. This Material Contains Barium Sulfate, a Contrast Dye That Makes it Easier to See the Port on Imaging. Unfortunately, Barium Sulfate Has Been Shown to Degrade the Structural Integrity of the Implant Itself. the Chemical Design Flaws in the PowerPort Catheters Have Caused the Device to Migrate, Deteriorate, and Fracture, Causing Significant Injury to Patients. the Migration and Deterioration Have Led to Devastating Outcomes for Many and Could Even Be Fatal if Left Untreated.
Do You Have a Bard PowerPort Catheter?
Due to Bard Refusing to Take Action to Recall This Product, Warn Consumers, or Modify Its Design for Safety, Numerous Lawsuits Have Been Filed. as of August 2023, the Cases Have Been Consolidated Into a Multidistrict Litigation. All Claimants Will Be Pursuing Bard Together in the U.S. District Court Before the Honorable David Campbell.
If You Have Experienced Any of the Following Symptoms or Complications From Your Bard PowerPort Catheter, Please Contact Your Doctor Immediately and Call the Neumann Law Group for an Evaluation of Your Claim:
- Infection/Sepsis
- Necrosis (Tissue Death)
- Vascular Perforation
- Organ Perforation
- Migration of Device Throughout the Body
- Fracture of the Device
- Internal Hemorrhage
- Heart Attack
- Stroke
- Cardiac Arrhythmia
- Pulmonary Embolism
- Blood Clots
- Hematoma
- Death
Do You Feel That You or a Loved One Have Been Injured as a Result of a Defective Bard PowerPort Catheter?
Neumann Law Group is Evaluating Claims Involving Those Who Have Received a Bard PowerPort Catheter and Have Experienced Any of the Above Symptoms/diagnoses. Neumann Law Group Takes Pride in Dedicating the Amount of Skill and Expertise to Bring Claims Against Pharmaceutical Companies. to Receive the Most Compensation for Your Injuries, Medical Expenses, Lost Wages, Pain and Suffering, and Other Damages You Should Be Represented by Educated and Involved Attorneys.
If You or Someone You Love Has Been Injured by Bard PowerPort Catheter, the Detroit, Grand Rapids, and Traverse City Defective Medical Device Lawyers at Neumann Law Group May Be Able to Help You Recover the Compensation You Deserve. We Represent Injured Patients in Petoskey, Warren, Holland, Midland, Muskegon, Saginaw, Wyoming, Kalamazoo, Lansing, Flint, Ann Arbor, and Communities Throughout the Upper Peninsula, as Well as in New York, California, and Massachusetts. Contact Us at 800-525-NEUMANN to Discuss Your Potential Case or Contact Us online.







