Internal Bra Mesh Lawsuit
Neumann Law Group is accepting claims against the manufacturers of internal bra mesh including AlloDerm, GalaFLEx, Phasix, and AlloMax regarding injuries sustained following breast reconstruction surgery due to complications from the mesh. Those who undergo breast reconstruction surgery, whether for cosmetic reasons or due to medical necessity, can experience sagging of the soft tissue under their implants. The internal bra mesh was designed as a supportive tool to prevent this sagging and mispositioning. Despite this type of mesh never being properly studied, tested, and approved by the Food and Drug Administration, manufacturers marketed it as safe to patients ultimately leading to significant complications including infection, mesh failure, breast deformity, significant pain, implant loss, or need for additional surgeries.
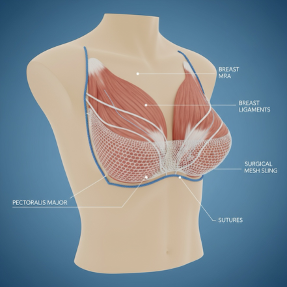
What Is Internal Bra Mesh?
An internal bra mesh is a built-in layer of support implanted by surgeons most often during breast surgery such as a lift, reduction, or reconstruction. Our bodies are designed so that skin and ligaments help to act like a “fabric”, holding up the soft tissue of the breast. Over time with aging and often with surgery, this soft tissue and ligaments begin to weaken. This internal bra mesh was designed to act as a second layer of support to hold everything in place, provide good shaping, and prevent sagging.
Internal bra mesh was placed on the market without first obtaining legitimate FDA approval. The internal bra mesh was only approved based on the fact that the manufacturers were able to show that there was already a marketed product that was substantially similar. This type of application. [501(k)] does not require any testing for safety or success and ultimately a very dangerous medical device was sold to doctors and patients. Marketing and sales of internal bra mesh was aggressive, and surgeons were encouraged to use the internal bra mesh without FDA approval solely to increase profits for these manufacturers. NO MESH PRODUCT HAS EVER BEEN APPROVED BY THE FDA FOR THE USE IN BREAST TISSUE! Despite this fact, bra mesh implants are still being used by surgeons regularly.
It took the FDA years to require these manufacturers to place labels on the internal bra mesh indicating that they were not studied or cleared for use in patients. Unfortunately, these warnings came too late and patients have suffered significant injury as a result.
As a result, legal claims are being evaluated regarding internal bra mesh claiming that this product with sold with knowledge of defects in design, manufacturing, and marketing defects. The legal claims also allege that the manufacturers of internal bra mesh have refused to admit the dangers of the mesh and continue to market it to patients and their doctors as safe and effective. There is still no indication of a formal recall.
The FDA should have performed a thorough evaluation of the internal bra mesh prior to approval. There is high pressure on patients to utilize these devices due to heavy marketing and claims of success. Patients and their physicians should be provided with accurate and non-deceptive information regarding their potential health outcomes. These manufacturers are driven by their profits, not by patient care.
Who May Be Eligible to Bring a Claim Against Internal Bra Mesh Manufacturers?
You may be eligible to file an internal bra mesh lawsuit if you or a loved one:
- Had breast reconstruction, augmentation, or revision surgery;
- Had internal bra mesh implant used in the procedure;
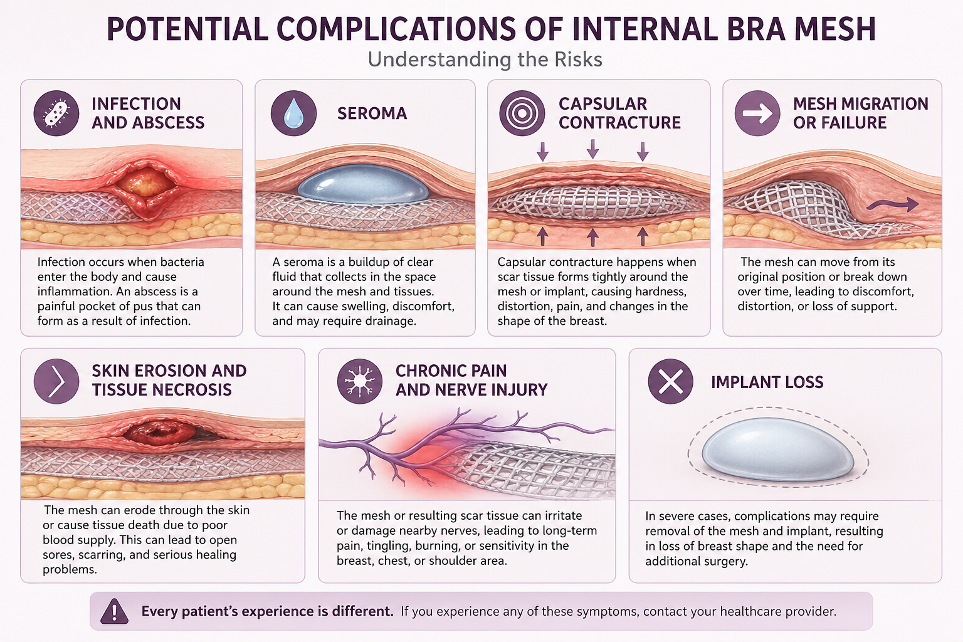
- Experienced complications after surgery including but not limited to infection and abscess, seroma, capsular contracture, mesh migration or failure, skin erosion and tissue necrosis, chronic pain and nerve injury, and implant loss
Do you feel that you or a loved one has been injured by a bra mesh implant?
Schedule a Free Consultation with Neumann Law Group’s Experienced Defective Medical Device Attorneys in Traverse City, Grand Rapids or Detroit
When one seeks relief cosmetic or reconstructive surgery, they typically take the word of a device manufacturer and their doctors regarding its safety. If the device manufacturer conceals information from governmental agencies and physicians, this trust can be broken, and people can be seriously injured. If your family finds itself in this challenging situation, Neumann Law Group’s attorneys maintain a wide range of skill and expertise in order to bring claims against product manufacturers. To receive the most compensation for your injuries, medical expenses, lost wages, pain and suffering, and other damages you should be represented by educated and involved attorneys.
Neumann Law Group is dedicated to serving our clients with a range of legal services including Mass Torts and Personal Injury throughout Michigan, as well as in New York, California, and Massachusetts. Contact us at 800-525-NEUMANN or via our online form for a free consultation with a product liability attorney.







